
Dutch Test
Testosterone Therapy in Males with Testosterone Deficiency (TD): Debunking the Myths
Since my last writing, I have continued my studies on female hormones and hormone replacement therapy and I have dug deep into the male literature on testosterone (T) and testosterone therapy (TTh). Similar to my quest to learn everything about female hormones and hormone therapy, the same holds true for testosterone and TTh in males.
In this writing, I am going to share some of what I have learned about T and TTh and provide you with the evidence to support your clinical decision making. This brief review will focus on debunking the myths associated with TTh. I chose this topic because, similar to menopausal hormone therapy (MHT) in females, there are misconceptions regarding TTh in testosterone deficient males (TD-M) that overshadow the evidence.
TD is associated with comorbidities that go beyond sexual symptoms. These include: metabolic syndrome, obesity, diabetes, hypertension, dyslipidemia, etc. Studies have documented increased mortality, greater atherosclerosis and coronary artery disease incidence, and increased low-trauma fracture rates.[i],[ii],[iii],[iv]
The evidence has consistently documented that TTh improves body composition, sexual function, bone mineral density, and metabolic parameters. In males with TD, TTh is not recommended to improve cognitive performance, even though low total testosterone (TT) levels are associated with cognitive decline. However, replacing TTh has not convincingly demonstrated cognitive improvement.[v],[vi],[vii],[viii],[ix],[x],[xi],[xii],[xiii],[xiv],[xv],[xvi],[xvii],[xviii],[xix],[xx],[xxi]
Despite TTh’s benefits, 2 concerns are often raised that may limit testosterone’s use in males who otherwise meet diagnostic criteria for TTh. These myths—TTh increases prostate cancer (PC) and increases cardiovascular disease (CVD)—will be addressed below. Let’s dig in to the data!
Truth: TTh does not cause PC
Since Huggins and Hodges reported in 1941 that castration led to metastatic PC regression and TTh administered to men with PC activates PC growth, the belief for decades had been that TTh causes PC. The evidence indicates that T has a limited ability to stimulate prostate growth with maximum androgen stimulation achieved at low TT concentrations. The saturation point, namely the TT concentration at which androgen binding to the androgen receptor is maximal, is approximately 250ng/dL.1-3,[xxii],[xxiii],[xxiv],[xxv],[xxvi]
The saturation model explains the paradoxical observation that prostate tissue is exquisitely sensitive to changes in serum TT at low concentrations, but becomes indifferent to changes at higher TT concentrations. A threshold effect occurs, in which increasing TT concentrations reach a limit (the saturation point) beyond which there is no further ability to induce androgen-driven changes in prostate tissue growth. See diagram below.1-3,22-26
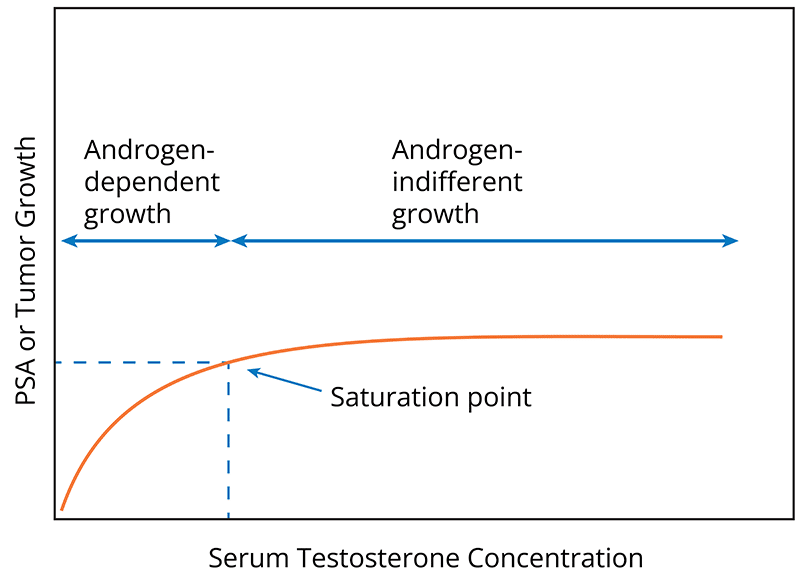
The saturation model shifted the paradigm away from the Huggins and Hodges’ androgen hypothesis. Despite this, Huggins and Hodges’ findings continue to raise concerns that TTh increases PC risk in males without a PC diagnosis.
This myth has been quashed by the extensive literature concluding that TTh does not increase PC risk.1-3,22-26,[xxvii],[xxviii] In 2004, Rhoden and Morgentaler26 published a comprehensive literature review and did not find compelling evidence that higher endogenous TT levels or TTh was associated with increased PC risk.26
In 2008, Roddam[xxix] published a frequently-cited collaborative analysis of pooled worldwide data from 18 longitudinal studies. Roddam found no association between PC risk and serum TT or dihydrotestosterone (DHT) concentrations. Males with higher TT levels were found to be at no greater PC risk than males with lower androgen levels. In other words, there is no significant relationship between serum androgens and PC risk.29
In 2012, Muller[xxx] published data from the REDUCE trial’s placebo arm. REDUCE was a randomized, double-blind, placebo-controlled, parallel-group study. Muller’s often-cited study documented that PC risk is unrelated to serum androgen concentrations. High TT levels do not predispose to PC and low TT levels are not protective.30,[xxxi]
The importance of Muller’s publication cannot be overemphasized. It lays to rest a false belief that has misinformed medical practice for decades. The truth is: T and TTh do not increase PC risk.
Since Muller’s publication, a large database has accumulated corroborating REDUCE’s placebo results. See appendix A, a table summarizing the often-cited key studies.

What about estradiol’s role in PC development? This is beyond the scope of this writing. Estrogens and the prostate have a complicated relationship. Prostatic estrogens are produced by androgen aromatization, either in-situ or peripherally. The prostate has both estrogen receptor alpha (ER-a) and estrogen receptor beta (ER-β). ER-β is the most prevalent estrogen receptor in the human prostate. The prostatic ratio of testosterone to estradiol and abnormal androgen aromatization to estrogens are 2 theories thought to be important for PC development and progression.[xxxvi],[xxxvii],[xxxviii],[xxxix],[xl],[xli]
Finally, in males on TTh who have an unexpected increase in PSA, don’t stop the TTh; just refer to a urologist.
Truth: There is no convincing evidence that TTh increases CV risk, non-fatal MI, or death
There are no long-term, large, randomized, double-blind, placebo-controlled trials evaluating TTh and cardiovascular outcomes. There is an ongoing large RCT (Testosterone Replacement Therapy for Assessment of Long-term Vascular Events and Efficacy in ResponSE in Hypogonadal Men [TRAVERSE]), which will address TTh and CVD.[xlii] What is promising is that the study has not been stopped early, suggesting the results are encouraging.
Despite no RCTs, a wealth of evidence has accumulated over the past 2 decades concluding: low serum TT levels are associated with increased atherosclerosis risk, CV risk factors, and mortality, and that TTh has beneficial effects on CV risk factors and risk biomarkers. The evidence supports TTh’s CV safety and that TTh may decrease CV events and mortality.[xliii],[xliv]
This all changed when 4 studies provided talking points for those who believed that TTh was unnecessary and TD didn’t really exist. Two studies were retrospective analysis (Vigen46 and Finkle47), 1 was a meta-analysis (Xu48), and there was 1 RCT T gel study (Basaria45). In 2014, these 4 studies led the FDA to mandate a label change to T products warning against possible MI and stroke. Men’s health experts all over the world have questioned these studies’ validity, accuracy, and credibility. See appendix B, a table summarizing these often-cited flawed studies.
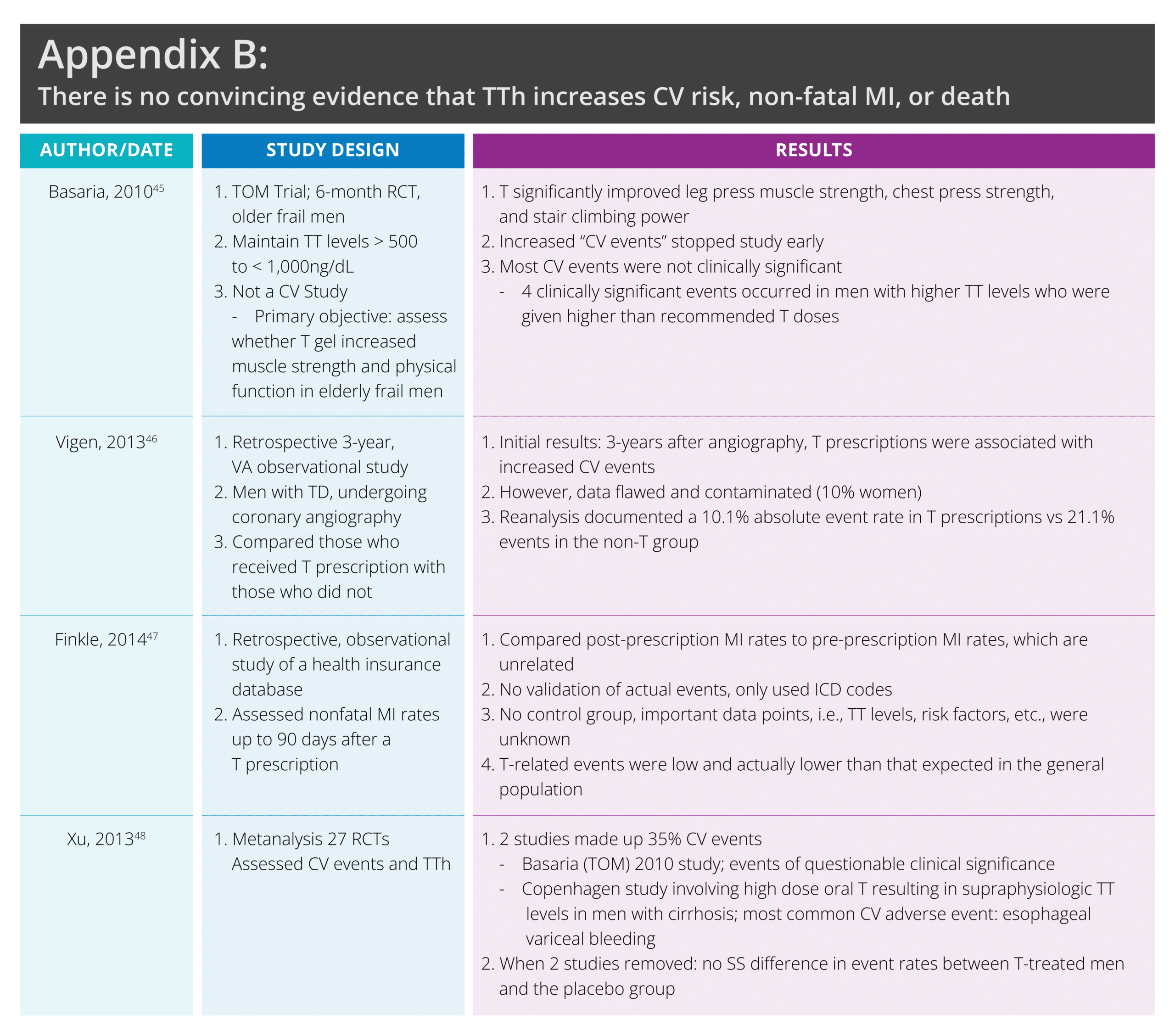
Vigen[xlvi] and Finkle[xlvii] were both retrospective analysis with methodological flaws. Vigen46 misrepresented its primary results and included 10% females in an all-male study. Vigen reported higher T-treated male events vs untreated males, when the absolute event rate was only 10.1% in the T-treated group vs. 21.2% in the untreated group.46
The Finkle47 study had serious data concerns. They compared pre-treatment MI rates with post-prescription MI rates, which measure different things. Further, T-related CV events were low and actually were lower than the expected rates in the general population.47
Xu’s[xlviii] metanalysis involving 27 studies found more CV events in T-treated males vs placebo-treated males. Two studies made up 35% of the cases. One was the TOM trial discussed above. The other was the 1986 Copenhagen study, where oral micronized T was given to males with cirrhosis. When these 2 studies are removed, the adverse CV event rates were similar in the T-treated and placebo groups, with a slightly lower rate in the T-group.48
Since the FDA advisory committee meeting in 2014, numerous studies have been published addressing CV risk and TTh. See appendix C, a table summarizing some often-cited studies documenting TTh’s safety and decreased event rates.
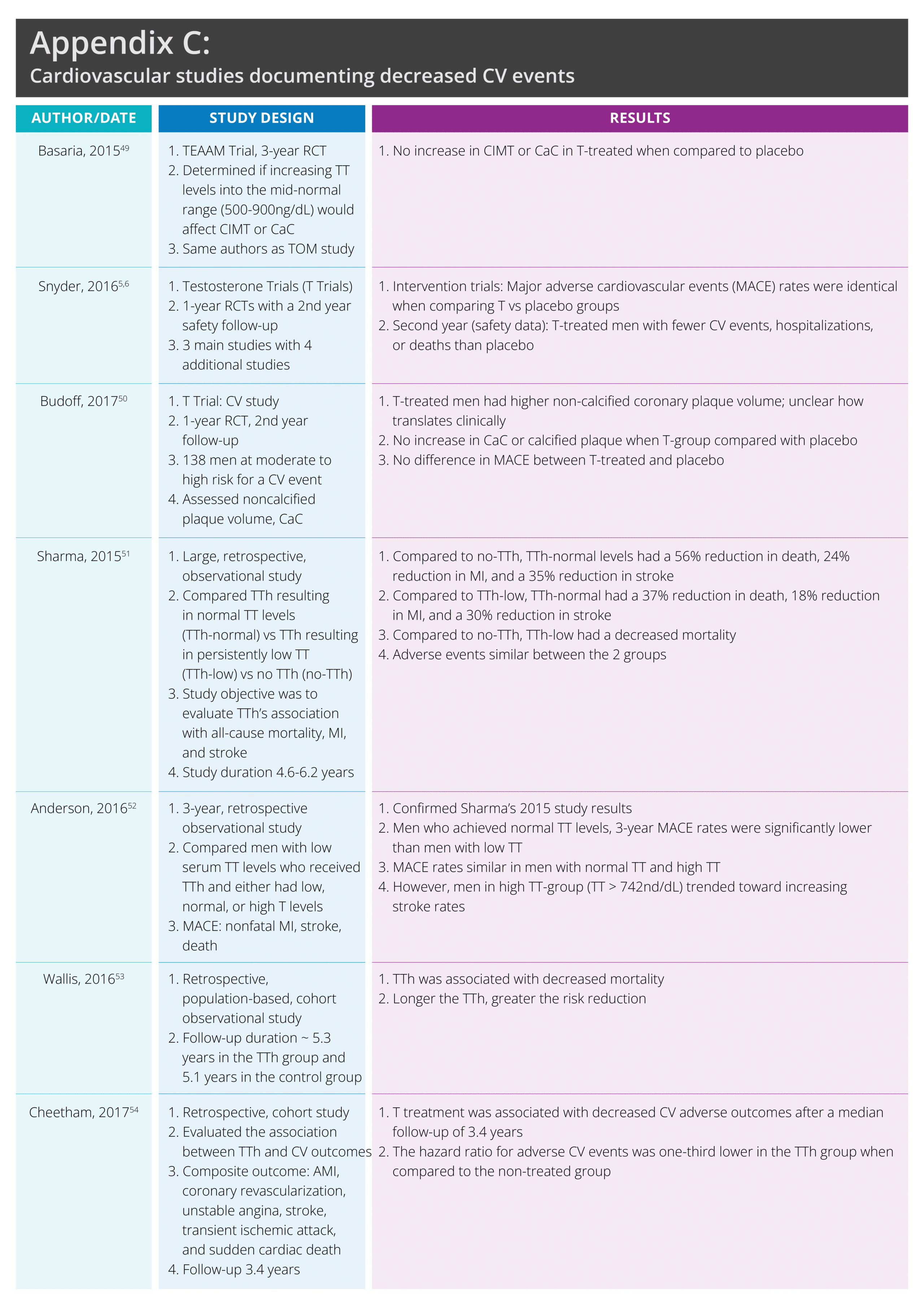
Sharma,[li] Anderson,[lii] Wallis,[liii] and Cheetham[liv] all documented that TTh was associated with decreased MACE rates. Sharma51 published a retrospective analysis concluding that MI and mortality were significantly decreased in males with TD, treated with TTh and whose serum TT levels normalized (> 500-800ng/dL), when compared to males who either received TTh but had persistently low TT levels or who received no TTh.51
Anderson52 confirmed Sharma’s results. Anderson52 reported that in males who achieved normal TT levels, the 3-year MACE and death rates were significantly lower when compared to males with low TT levels (< 212ng/dL). MACE rates were similar for males with higher TT levels and normal TT levels. However, in males with high TT levels (> 742ng/dL) there was a trend towards increased stroke risk. The authors recommended a conservative approach to TTh.52
Wallis53 published a population-based, matched-cohort study assessing the association between cumulative TTh exposure and mortality, CV events, and PC. Wallis documented that the longer the TTh, the greater the mortality risk reduction.53
Cheetham54 found that TTh was associated with decreased MACE after a median follow-up of 3.4 years. The hazard ratio for adverse CV events was one-third lower in the T-treated group when compared to the non-treated group.54
Conclusion
TTh in males with TD is safe and does not increase PC. There is a TT saturation point (TT ~ 250ng/dL) where there is no further ability to induce androgen-driven changes in prostate tissue growth. Estradiol’s role in PC is complicated. The literature suggests that the prostatic ratio of testosterone to estradiol and aberrant androgen aromatization to estrogens are 2 theories thought to be important for PC development and progression.
The evidence suggesting that TTh increases CV risk is weak and flawed. The International Expert Consensus Conference on Testosterone Deficiency and its Treatment,2 along with its 2019 update,[lv] and the American Association of Clinical Endocrinologists[lvi] all conclude: the evidence does not indicate increased CV risk with TTh.
Notwithstanding all the evidence I presented, before prescribing TTh, I would recommend CV risk stratification. In addition, all males being prescribed TTh should be counseled regarding the black-box warning that is on all T prescriptions. I hope I have provided sound evidence to support TTh’s safe use in males and to not hold back necessary T prescriptions because of 4 flawed studies that received a lot of press.
[i] Morgentaler A, Traish AM. The history of Testosterone and the Evolution of its Therapeutic Potential. Sex Med Rev. 2020; 8(2): 286-296.
[ii] Morgentaler A, et al. Fundamental Concepts Regarding Testosterone Deficiency and Treatment: International Expert Consensus Resolutions. Mayo Clin Proc. 2016; 91(7): 881-896.
[iii] Traish AM. Benefits and Health Implications of Testosterone Therapy in Men With Testosterone Deficiency. Sex Med Rev. 2018; 6(1): 86-105.
[iv] Traish A. Testosterone therapy in men with testosterone deficiency: are the benefits and cardiovascular risks real or imagined? Am J Physiol Regul Integr Comp Physiol. 2016; 311(3): R566-573.
[v] Snyder PJ, et al. Lessons from the Testosterone Trials. Endocr Rev. 2018; 39(3): 369-386.
[vi] Snyder PJ, et al. Effects of Testosterone Treatment in Older Men. N Engl J Med. 2016; 374(7): 611–624.
[vii] Wang C, et al. Transdermal Testosterone Gel Improves Sexual Function, Mood, Muscle Strength, and Body Composition Parameters in Hypogonadal Men. J Clin Endocrinol Metab. 2000; 85(8): 2839-2853.
[viii] Wang C, et al. Long-Term Testosterone Gel (AndroGel) Treatment Maintains Beneficial Effects on Sexual Function and Mood, Lean and Fat Mass, and Bone Mineral Density in Hypogonadal Men. J Clin Endocrinol Metab. 2004; 89(5): 2085-2098.
[ix] Cunningham GR, et al. Testosterone Treatment and Sexual Function in Older Men With Low Testosterone Levels. J Clin Endocrinol Metab. 2016 Aug;101(8):3096-3104.
[x] Corona G, et al. Meta-analysis of Results of Testosterone Therapy on Sexual Function Based on International Index of Erectile Function Scores. Eur Urol. 2017; 72(6): 1000-1011.
[xi] Brock G, et al. Effect of Testosterone Solution 2% on Testosterone Concentration, Sex Drive and Energy in Hypogonadal Men: Results of a Placebo Controlled Study. J Urol. 2016; 195(3): 699-705.
[xii] Mohamad NV, et al. A concise review of testosterone and bone health. Clin Interv Aging. 2016 Sep;11: 1317-1324.
[xiii] Golds G, et al. Male Hypogonadism and Osteoporosis: The Effects, Clinical Consequences, and Treatment of Testosterone Deficiency in Bone Health. Int J Endocrinol. 2017; 2017: 4602129.
[xiv] Haider A. Progressive Improvement of T-Scores in Men with Osteoporosis and Subnormal Serum Testosterone Levels upon Treatment with Testosterone over Six Years. Int J Endocrinol. 2014; 2014: 496948.
[xv] Snyder PJ, et al. Effect of Testosterone Treatment on Volumetric Bone Density and Strength in Older Men With Low Testosterone: A Controlled Clinical Trial. JAMA Intern Med. 2017; 177(4): 471-479.
[xvi] Traish AM, et al. Long-Term Testosterone Therapy Improves Cardiometabolic Function and Reduces Risk of Cardiovascular Disease in Men with Hypogonadism: A Real-Life Observational Registry Study Setting Comparing Treated and Untreated (Control) Groups. Cardiovasc Pharmacol Ther. 2017; 22(5): 414-433.
[xvii] Hsu B, et al. Longitudinal Relationships between Reproductive Hormones and Cognitive Decline in Older Men: The Concord Health and Ageing in Men Project. Clin Endocrinol Metab. 2015; 100(6): 2223-2230.
[xviii] Huang G, et al. Effects of long-term testosterone administration on cognition in older men with low or low-to-normal testosterone concentrations: a prespecified secondary analysis of data from the randomised, double-blind, placebo-controlled TEAAM trial. Lancet Diabetes Endocrinol. 2016; 4(8): 657-665.
[xix] Kenny AM, et al. Effects of Testosterone on Behavior, Depression, and Cognitive Function in Older Men With Mild Cognitive Loss. J Gerontol A Biol Sci Med Sci. 2004; 59(1): 75-78.
[xx] Cherrier MM, et al. Testosterone Treatment of Men With Mild Cognitive Impairment and Low Testosterone Levels. Am J Alzheimers Dis Other Demen. 2015; 30(4): 421-430.
[xxi] Resnick SM, et al. Testosterone Treatment and Cognitive Function in Older Men With Low Testosterone and Age Associated Memory Impairment. JAMA. 2017; 317(7): 717-727.
TTh and PC
[xxii] Diagnosis and Treatment of Testosterone Deficiency: Updated Recommendations From The Lisbon 2018 International Consultation for Sexual Medicine. Sex Med Rev. 2019; 74(4): 636-649.
[xxiii] Morgentaler A, et al. A Critique of the AUA Guidelines. J Sex Med. 2020; 17(4): 561-564.
[xxiv] Morgentaler A, Traish AM. Shifting the Paradigm of Testosterone and Prostate Cancer: The Saturation Model and the Limits of Androgen-Dependent Growth. Eur Urol. 2009; 55(2): 310-320.
[xxv] Khera M, et al. A New Era of Testosterone and Prostate Cancer: From Physiology to Clinical Implications. Eur Urol. 2014; 65(1): 15-23.
[xxvi] Rhoden El, Morgentaler A. Risks of Testosterone-Replacement Therapy and Recommendations for Monitoring. N Engl J Med. 2004; 350(5): 482-492.
[xxvii] Morgentaler A. Goodbye Androgen Hypothesis, Hello Saturation Model. Eur Urol. 2012; 62(5): 765-767.
[xxviii] Cui Y, et al. The effect of testosterone replacement therapy on prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2014; 17(2): 132-143.
[xxix] Roddam AW, et al. Endogenous Hormones and Prostate Cancer Collaborative Group. Endogenous Sex Hormones and Prostate Cancer: A collaborative Analysis of 18 Prospective Studies. J Natl Cancer Inst. 2008; 100(3): 170-183.
[xxx] Muller RL, et al. Serum Testosterone and Dihydrotestosterone and Prostate Cancer Risk in the Placebo Arm of the Reduction by Dutasteride of Prostate Cancer Events Trial. Eur Urol. 2012; 62(5): 757-764.
[xxxi] Andriole GL, et al. Effect of Dutasteride on the Risk of Prostate Cancer. N Engl J Med. 2010; 362(13): 1192-1202.
[xxxii] Loeb S, et al. Testosterone Replacement Therapy and Risk of Favorable and Aggressive Prostate Cancer. J Clin Oncol. 2017; 35(13):1430-1436.
[xxxiii] Baillargeon J, et al. Long-term Exposure to Testosterone Therapy and the Risk of High Grade Prostate Cancer. J Urol. 2015; 194(6): 1612-1616.
[xxxiv] Schenk JM, et al. Serum androgens and prostate cancer risk: results from the placebo arm of the Prostate Cancer Prevention Trial. Cancer Causes Control. 2016; 27(2): 175-182.
[xxxv] Wallis CJD, et al. Survival and cardiovascular events in men treated with testosterone replacement therapy: an intention-to-treat observational cohort study. Lancet Diabetes Endocrinol. 2016; 4(6): 498-506.
[xxxvi] Cooke PS, et al. Estrogens In Male Physiology. Physiol Rev. 2017; 97(3): 995-1043.
[xxxvii] Dobbs RW, et al. Estrogens and Prostate Cancer. Prostate Cancer Prostatic Dis. 2019; 22(2): 185-194.
[xxxviii] Christoforou P, et al. The role of estrogen receptor β in prostate cancer. Mol Med. 2014; 29(1): 427-434.
[xxxix] Bonkhoff H. Estrogen receptor signaling in prostate cancer: Implications for carcinogenesis and tumor progression. Prostate. 2018; 78(1): 2-10.
[xl] Di Zazzo E, et al. Estrogens and Their Receptors in Prostate Cancer. Therapeutic Implications. Front. Oncol. 2018; 8: 2.
[xli] Bozovic A, et al. Estrogen Receptor Beta: The Promising Biomarker and Potential Target in Metastases. Int J Mol Sci. 2021; 22(4): 1656.
TTh and Cardiovascular Disease
[xlii] Bhasin S, et al. Effects of long-term testosterone treatment on cardiovascular outcomes in men with hypogonadism: Rationale and design of the TRAVERSE study. Am Heart J. 2022; 245: 41-50.
[xliii] Morgentaler A, et al. Testosterone Therapy and Cardiovascular Risk: Advances and Controversies. Mayo Clin Proc. 2015; 90(2): 224-251.
[xliv] Miner M, et al. The state of testosterone therapy since the FDA’s 2015 labelling changes: Indications and cardiovascular risk. Clin Endocrinol (Oxf). 2018; 89(1): 3-10.
[xlv] Basaria S, et al. Adverse Events Associated with Testosterone Administration. N Engl J Med. 2010; 363(2): 109-122.
[xlvi] Vigen R, et al. Association of Testosterone Therapy With Mortality, Myocardial Infarction, and Stroke in Men With Low Testosterone Levels. JAMA. 2013; 310(17): 1829-1836.
[xlvii] Finkle WD, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014; 9(1): e85805.
[xlviii] Xu L, et al. Testosterone therapy and cardiovascular events among men: a systematic review and meta-analysis of placebo-controlled randomized trials. BMC Med. 2013; 11: 108.
[xlix] Basaria S, et al. Effects of Testosterone Administration for 3 Years on Subclinical Atherosclerosis Progression in Older Men With Low or Low-Normal Testosterone Levels. JAMA. 2015; 314(6): 570-581.
[l] Budoff MJ, et al. Testosterone Treatment and Coronary Artery Plaque Volume in Older Men With Low Testosterone. JAMA. 2017; 317(7): 708-716.
[li] Sharma R, et al. Normalization of testosterone level is associated with reduced incidence of myocardial infarction and mortality in men. Eur Heart J. 2015; 36(40): 2706-2715.
[lii] Anderson JL, et al. Impact of Testosterone Replacement Therapy on Myocardial Infarction, Stroke, and Death in Men With Low Testosterone Concentrations in an Integrated Health Care System. Am J Cardiol. 2016; 117(5): 794-799.
[liii] Wallis CJ, et al. Survival and cardiovascular events in men treated with testosterone replacement therapy: an intention-to-treat observational cohort study. Lancet Diabetes Endocrinol. 2016; 4(6): 498-506.
[liv] Cheetham TC. Association of Testosterone Replacement With Cardiovascular Outcomes Among Men With Androgen Deficiency. JAMA Intern Med. 2017; 177(4):491-499.
[lv] Morgentaler A, et al. Diagnosis and Treatment of Testosterone Deficiency: Updated Recommendations From the Lisbon 2018 International Consultation for Sexual Medicine. Sex Med Rev. 2019; 7(4): 636-649.
[lvi] Bhasin S, et al. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2018; 103(5): 1715-1744.

By: Doreen Saltiel, MD, JD
Doreen Saltiel, MD, JD, has practiced medicine for almost 40 years. She completed a cardiologyfellowship and board certification and practiced interventional cardiology for more than 25 years.She currently practices functional medicine with an emphasis on hormone health in both men andwomen as well as preventive cardiology. Dr. Saltiel completed advanced fellowship training inMetabolic and Nutritional Medicine from MMI and is a Diplomate of the American Board of Anti-Aging and Regenerative Medicine. She also completed MMI’s advanced certification inEndocrinology and Cardiovascular Health. Dr. Saltiel has co-authored multiple peer-reviewedpapersand recently joined the Founder of Precision Analytical, Mark Newman, to present hormone testingresearch at the North American Menopause Society’s Annual Meeting








