Click here for more information on the Doctor’s Data GI360 stool profile, including downloadable resource guide, sample reports, FAQs and more!
Contact info: info@doctorsdata.com
In recent years, glucagon-like peptide-1 receptor agonists (GLP-1 RAs) use has increased 700% in the general population and over one quarter of those diagnosed with diabetes are on these medications.
Recent studies have demonstrated that GLP-1 RAs exert anti-inflammatory effects, expanding their therapeutic use beyond glycemic control. These findings introduce a relevant consideration within functional and longevity medicine, where modulation of systemic inflammation is paramount. However, these benefits may never come to fruition if GLP-1 RAs induce dietary modifications that create maladaptive gut microbiome shifts, especially over the long term. Appetite suppression and compromised nutrient intake associated with GLP-1 RAs can reduce gut microbiome diversity, induce dysbiosis, and subsequently increase inflammation. In these cases, the anti-inflammatory and metabolic advantages may be offset.
The increased prevalence of these medications, alongside their physiological effects, underscores the need for clinicians to expect to see an increasing number of patients on these medications, even without diabetes, and be prepared to evaluate and manage the effects of GLP-1 RAs.
Secondary Effects of GLP-1 Agonists: Acute GI Symptoms vs Chronic Gut Microbiome Dysbiosis
Some secondary effects of GLP-1 RAs are symptomatic and relatively acute, such as gastrointestinal (GI) changes (constipation, diarrhea, nausea and vomiting), therefore mitigation strategies are well known. Other secondary effects of GLP-1 RAs, such as gut microbiome shifts, may be asymptomatic and chronic with far-reaching physiologic potential. These secondary effects, such as gut microbiome dysbiosis, reduced diversity, and barrier compromise, may offset the anti-inflammatory effects of these medications.

Fig. 1: GI Health Markers on the Doctor’s Data GI360 provides a summary of functional guilds of bacteria and allows personalized clinically actionable insight. GLP-1 RA-induced appetite suppression and unstructured reduced dietary intake can significantly influence bacterial populations, leading to GI health markers outside the reference interval.
Why Chronic Microbiome Shifts Often Go Unaddressed
There exists an overlap between transient GI symptoms and chronic gut microbiome shifts, while the former receives clinical attention, and the latter remains largely unaddressed. From a clinical perspective, it is relevant to address gut microbiome composition as this is a foundational component of human physiology. The gut microbiome is fundamental to biological processes, including nutrient absorption, drug metabolism, energy homeostasis, immunity, and autoimmunity.
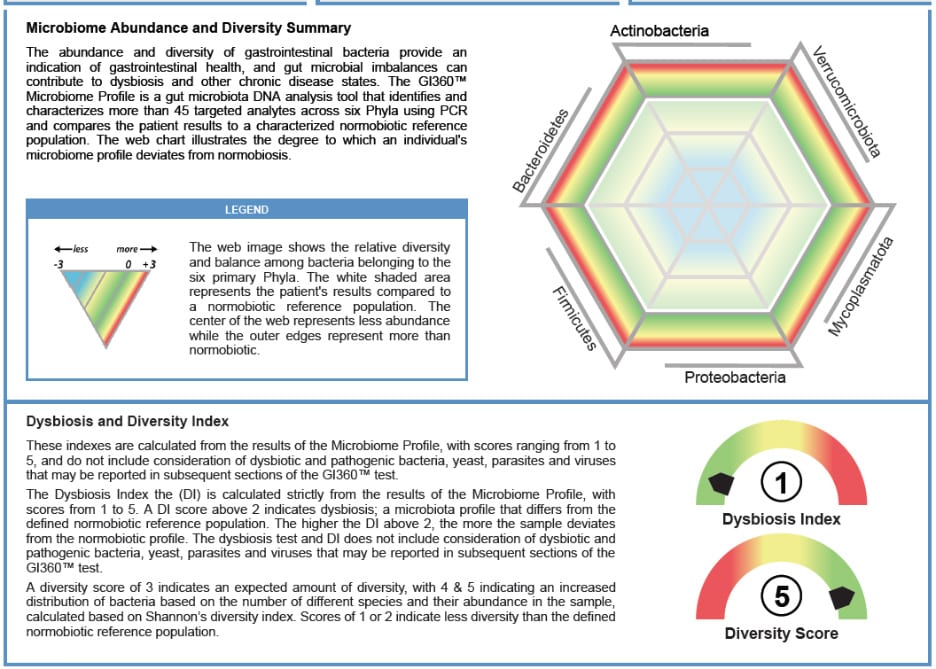
Fig. 2: GI360™ is a powerful tool to profile the gut microbiome via PCR analysis of abundance, diversity and dysbiosis. The web represents microbiome abundance and diversity compared to a published normobiotic reference population. A healthy microbiome is represented by a hexagonal shape, indicating balance and adequate bacterial populations. Deviations from normobiosis may be represented by various shapes, square, triangle, etc., and various sizes of the web. Smaller webs indicate lower gut microbiome populations. The Dysbiosis Index (DI) is a calculation with scores from 1 to 5 based on the overall bacterial abundance and profile within the patient’s sample as compared to a reference population. Values above 2 indicate a microbiota profile that differs from the defined normobiotic reference population (i.e., dysbiosis). The higher the DI above 2, the more the sample is considered to deviate from normobiosis. A diversity score of 3 and above indicates an expected amount of diversity. In clinical practice, this helps bridge complex gut microbiome data, systemic health, and individualized treatment considerations, supporting a shared understanding of gut-driven mechanisms, and improving patient outcomes.
Gut Microbiome Dysbiosis, Inflammation, and Systemic Risk
Subsequently, gut microbiome dysbiosis has been associated with metabolic dysfunction, poor mental health, chronic disease, cardiovascular disease (CVD), reduced longevity, neurodegeneration, and inflammation.
Regarding secondary effects of GLP-1 RAs, anti-inflammatory benefits emerge as the biochemical pathways are elucidated. GLP-1 RAs regulate immune cell signaling, suppress proinflammatory cytokines (including tumor necrosis factor alpha (TNFα), interleukin-6 (IL-6), and interleukin-1 beta (IL-1β)), enhance anti-inflammatory cytokines (interleukin-10- (IL-10)), and decrease reactive oxygen species. They blunt activation of nuclear factor kappa B (NF–κB), reducing transcription of inflammatory mediators, and promote anti-inflammatory signaling and metabolic equilibrium via AMP-activated kinase (AMPK) activation.
Together, these secondary effects improve metabolism and also confer protection against chronic inflammation. These effects may prove promising for not only Type 2 Diabetes (T2D) and obesity, but also CVD, neurodegenerative disorders (Alzheimer’s, Parkinson’s), and Inflammatory Bowel Disease (IBD).
It has been noted that the primary weight reduction effects of GLP-1 RAs may be attributed to neurobiological appetite suppression and subsequent reduced energy intake. A clinical trial found that individuals on 50mg of oral semaglutide weekly decreased their energy intake by 39%. This effect is often the desired outcome for those taking this medication, yet additional considerations exist in practice and warrant clinical management. A reduction in caloric intake without structured dietary guidance can lead to unintended adverse effects on the gut microbiome.
How GLP-1 Agonists Influence the Gut Microbiome: Evidence vs Real-World Diet Patterns
What Preclinical Models Suggest About GLP-1 RAs and Microbial Diversity
A publication examining the effects of GLP-1 RAs on the gut microbiome, primarily in murine models, reports beneficial shifts in microbial composition. Gut microbial effects include improved diversity, reduction in inflammatory bacteria, and an increase in the following keystone bacteria: Faecalibacterium prausnitzii (F. Prausnitzii), Lactobacillus spp., Akkermansia muciniphila (A. muciniphila).These positive changes support mucosal barrier integrity and endogenous glucagon-like peptide-1 (GLP-1) production leading to a subsequent improvement in insulin sensitivity. This evidence is consistent with the anti-inflammatory effects of these medications. Such a direct conclusion cannot be drawn with humans, as the quality of the diet is as relevant as quantity with regards to the gut microbiome.
Appetite Suppression, Diet Quality, and Nutrient Dilution in Clinical Practice
In clinical practice, GLP-1 RAs suppress appetite so effectively that many patients report not eating for most of the day. When hunger finally breaks through, it often manifests as cravings for caloric-dense, nutrient-deficient, palatable foods (pizza, cake, fried foods, ice cream, etc.). The Standard American Diet (SAD) compounds this GLP-1 RAs conundrum, as individuals may be primarily consuming nutrient deficient foods.
Nutrient Deficiency and Emerging Gut Microbiome Risks
Even if individuals are aware of the physiological impact of food items, they may justify nutrient deficient food choices due to the GLP-1 RA-induced prolonged fasting periods. This GLP-RA dietary pattern leads to decreased intake of key nutrients such as fiber, protein, vitamins, and minerals, often below physiological needs. Observed risks include malnutrition, specifically thiamine deficiency, metabolic acidosis, and hypomagnesemia. Over time, these dietary choices can compound into patterns that adversely impact the gut microbiome, which may influence physiology to negate the beneficial effects of the GLP-1 RAs. This phenomenon underscores the need for clinicians to monitor nutritional status and gut microbiome biomarkers.
Gut Microbiome Risks of Unstructured Reduced Dietary Intake
Restrictive eating patterns inadvertently disrupt physiological homeostasis, including the gut microbiome. The absence of nutrient diversity, imbalanced macronutrient intake and decreased phytochemical intake can lead to a loss of gut microbial diversity, including keystone bacteria, and simultaneously increased growth of opportunistic species, contributing to subsequent dysbiosis, reduced short-chain fatty acid (SCFAs) production, and inflammation.
Diversity
Gut microbiome diversity is widely recognized as a biomarker of health. Higher diversity in gut microbial communities is associated with resilience, metabolic balance, and reduced risk of chronic disease. On the flip side reduced diversity is linked to inflammation, metabolic disorders, and impaired immune function.
Increasing gut microbiome diversity through a diet rich in fermented foods leads to a measurable reduction in systemic inflammation. A landmark study demonstrated gut microbiome diversity decreased activation across four major immune cell types (CD4+ T cells, CD8+ T cells, B cells, and monocytes), alongside reductions in 19 circulating inflammatory proteins, including IL-6. These findings indicate a broad downregulation of immune activation, suggesting improved immune tolerance and resilience, potentially conferring benefits regarding the pathophysiology associated with arthritis, T2D, and chronic stress. In essence, enhanced microbial diversity functions as a mediator that dampens proinflammatory cytokine signaling, thereby supporting proper immune regulation and reduced risk of chronic disease.
It has been consistently observed that gut microbiome diversity declines with age, and this may be associated with the pathophysiology of senescence. In Blue Zone populations, gut microbiome diversity is maintained well into old age, acting as a mediator between environmental exposures, immune and metabolic health, thereby positively influencing inflammation and cellular aging processes.
Restrictive eating patterns inherently low in a variety of fiber, resistant starches, and polyphenol-rich foods diminish substrates required for diverse gut bacterial populations and adequate microbiome diversity. Individuals engaging in unstructured reduced dietary intake while on GLP-1 RAs should be monitored for gut microbiome diversity, as preserved diversity is critical for maintaining the anti-inflammatory effects associated with a healthy microbiome.

Fig. 3: This example reflects GLP-1 RAs unstructured reduced dietary intake and a patient struggling to maintain adequate fiber. The GI360™ Microbiome Profile is a focused gut microbiota DNA analysis tool that identifies targeted analytes using a CE-marked multiplex PCR system. Individual results are compared to a highly defined normobiotic reference population. A Dysbiosis Index (DI) of 5 is associated with IBD, IBS, obesity, metabolic dysfunction, and observed in autoimmunity. A low Diversity Score (DS) of 2, is associated with adverse health outcomes. This report provides personalized actionable findings and allows for patient education via illustrated gut microbiome biomarkers.
Dysbiosis
Dysbiosis, defined as reduced diversity, loss of beneficial microbes, or overgrowth of pathogenic species, is associated with conditions such as irritable bowel syndrome (IBS), IBD, obesity, diabetes, and depression. Dysbiosis may develop due to a decline in beneficial taxa, while opportunistic species increase. Unstructured reduced dietary intake is consistent with caloric-dense, nutrient-deficient, palatable foods that do not provide substrates, such as fiber and polyphenols, to support beneficial bacteria, such as Bifidobacterium, A. muciniphila, Lactobacillus spp., and support the growth of potentially inflammatory bacteria, such as Proteobacteria.

Fig. 4: Bifidobacterium, Akkermansia muciniphila, and Lactobacillus spp. are keystone bacteria identified in GI360™. Unstructured reduced dietary intake during GLP-1 RA therapy may lower levels of these keystone bacteria, with potentially significant and wide-ranging physiological consequences. Objective assessment of these key bacteria allows for precise clinical action regarding GI health, gut microbiome balance, mucosal integrity, immune balance, metabolic health, and more. These values may be utilized to guide and monitor therapeutics such as diet and probiotics.
These changes in the gut microbiome lead towards a predisposition to decreased SCFA production, compromised mucosal barrier integrity and subsequent gut permeability, allowing lipopolysaccharides (LPS) circulation, fueling chronic low-grade inflammation. Gut microbial dysbiosis alters regulatory T cells (Tregs) and cytokine balance, perpetuating inflammatory pathways even in the absence of overt infection. Clinical sequelae include CVD, metabolic disorders, and neurocognitive disorders as dysbiosis is associated with “inflammaging”, the chronic low-grade inflammation that accelerates atherosclerosis, insulin resistance and cognitive decline.
Patients undergoing GLP-1 RA therapy who adopt unstructured reductions in dietary intake should be closely monitored for gut microbiome dysbiosis, as preservation of gut microbiome eubiosis is foundational to health. Evaluation of the levels and proportional relationships among key commensal taxa, including Bifidobacterium, A. muciniphila, and Lactobacillus spp., is recommended, alongside monitoring for the expansion of potentially proinflammatory organisms such as Proteobacteria. Addressing these imbalances is essential to preserve normobiosis and mitigate downstream inflammatory risk.

Fig. 5: GI360™ includes inflammatory biomarkers such as lactoferrin, lysozyme, and calprotectin, alongside the immunology marker fecal secretory immunoglobulin A (sIgA). These biomarkers help to distinguish IBD from IBS and garner insight into immune health of the gut microbiome. They primarily reflect inflammation and mucosal immune activity, which may occur with microbial imbalance.
Short Chain Fatty Acids (SCFAs)
Unstructured reduced dietary intake is often associated with lower intake of fermentable fibers (found in onion, leeks, asparagus, legumes, oats, cooled oats and rice, apples, pears, carrots, etc.). Decreased intake of fermentable fibers contributes to decreased microbiome diversity, a decline in beneficial taxa, such as Bifidobacterium, F. prausnitzii and Lactobacillus spp., which diminishes short chain fatty acid (SCFA) production, weakens intestinal barrier integrity, and increases the risk of dysbiosis and systemic inflammation. SCFAs, such as acetate (C2), propionate (C3), and butyrate (C4), are critical for epithelial barrier integrity, and anti-inflammatory signaling.Low levels are associated with IBD, cancer, obesity, diabetes, kidney disease, hypertension, liver disease, immune system dysfunction, and neurocognitive conditions.

Fig. 6: Fecal short-chain fatty acids (SCFAs) are microbial metabolites that serve as key indicators of gut health and integrity. Their measurement in stool provides a non-invasive biomarker associated with microbiome function to guide interventions and evaluate therapeutic responses in gastrointestinal disease management. SCFAs are often imbalanced in patients with inadequate fiber intake, including diets influenced by GLP-1 RAs.
Butyrate is the primary energy source for colonocytes, as it strengthens tight junction proteins (zona occludens-1 (ZO-1) and claudins), reducing intestinal permeability. SCFAs inhibit NF-κB activation and promote differentiation of Tregs via histone deacetylase inhibition, enhancing immune tolerance. Butyrate and propionate reduce systemic inflammation by lowering IL-6 and T-NFα.
F. prausnitzii is the most abundant butyrate producing bacteria in a healthy gastrointestinal tract and protective factor for the intestinal mucosa.

Fig. 7: Diets influenced by GLP-1 RAs often lead to imbalances in key bacteria like F. prausnitzii and A. muciniphila leading to reduced mucosal barrier integrity and increased intestinal permeability. The summary table included in the Doctor’s Data GI360TM Resource Guide highlights Functional Markers, ie. key mucosa-protective bacteria and flags when their levels fall outside healthy thresholds. This organization links clinical interpretation of stool profiles to microbial imbalance and health implications, allowing targeted interventions.
Under the influence of dietary restriction, a robust population of F. prausnitzii is particularly at risk, as this bacterium is largely dependent upon adequate dietary consumption of fermentable soluble fiber, found in legumes, fruits, vegetables, and whole grains. Decreased levels of F. prausnitzii contribute to dysbiosis, increased gut permeability, systemic inflammation and are associated with IBD, IBS, celiac disease, Parkinson’s disease, bipolar disorder, colorectal cancer and diabetes. Monitoring levels of F. prausnitzii and SCFAs is recommended to support individuals undergoing GLP-1 RA therapy associated with decreased dietary intake.
Current patient education on GLP-1 RAs emphasizes adequate protein intake, primarily to preserve muscle mass, while giving less attention to dietary fiber, despite its important roles in satiety, glycemic control, and gut microbiome health. Because of its wide-ranging systemic effects, many mediated by gut microbial metabolites such as short-chain fatty acids (SCFAs), adequate dietary fiber intake is essential for maintaining homeostasis. However, diet diaries provide limited clinical insight due to their subjective nature, and this information is often underweighted relative to the profound physiological impact of the gut health axis. Therefore, incorporating objective gut microbiome biomarkers represents best practice in the clinical setting to generate actionable insights.
GLP-1 RA Therapy & Multidisciplinary Care: Gut Microbiome Monitoring
For individuals on GLP-1 RAs stool testing for diversity, dysbiosis, keystone bacteria, and SCFAs provides an individualized biomarker panel that links microbiome composition to inflammation, metabolic resilience, and intestinal health. This multidisciplinary approach ensures that reduced dietary intake from GLP-1 RA therapy does not inadvertently compromise microbiome composition, which is essential for sustaining the anti-inflammatory benefits of treatment.
Stool analysis can identify gut microbiome diversity and dysbiosis, loss of beneficial taxa, or overgrowth of proinflammatory organisms. Assessing both the abundance and relative balance of core commensal organisms, such as Bifidobacterium, A. muciniphila, F. prausnitzii, and Lactobacillus spp., is advisable, with parallel attention to the overgrowth of proinflammatory taxa like Proteobacteria. Timely recognition and correction of these shifts are critical for maintaining normobiosis and reducing the risk of chronic inflammatory sequelae. Measurement of SCFAs in stool reflects microbial metabolic activity, with butyrate, acetate, and propionate supporting immune tolerance and anti-inflammatory signaling.
Doctor’s Data GI360™ provides individualized profiling of the gut microbiome including diversity, dysbiosis, keystone bacteria and SCFAs. GI360™ is a gut microbiota DNA analysis tool that identifies and characterizes the abundance and diversity of more than 45 targeted analytes that peer-reviewed research has shown to contribute to dysbiosis and other chronic disease states.

Fig. 8: The GI360™ case example here denotes a balanced web with a WNL Dysbiosis Index (DI) of 2 and a Diversity Score of 4. The normobiotic microbiome and healthy diversity reflects the patient’s reported diet, which is high in soluble fiber, polyphenols, and fermented foods. This equates to adequate butyrate secretion and low inflammation. In practice, GI360™ supports personalized treatment strategies, dietary interventions, and monitoring of therapeutic outcomes, making it a valuable tool for patient education and clinical outcomes.
The Dysbiosis Index (DI) is a calculation with scores from 1 to 5 based on the overall bacterial abundance and profile within the patient’s sample as compared to a reference population. Values above 2 indicate a microbiota profile that differs from the defined normobiotic reference population (i.e., dysbiosis). The higher the DI above 2, the more the sample is considered to deviate from normobiosis. A diversity score of 3 indicates an expected amount of diversity, with 4 & 5 indicating an increased distribution of bacteria based on the number of different species and their abundance in the sample, calculated based on Shannon’s diversity index. Scores of 1 or 2 indicate less diversity than the defined normobiotic reference population. Objective values of Bifidobacterium, A. muciniphila, F. prausnitzii, and Lactobacillus spp. are included in the GI360™ for individualized insight into these keystone bacteria.
GLP-1 RAs effectively reduce appetite and caloric intake, yet unstructured reduced dietary intake is often nutrient poor and patients risk losing the fiber, polyphenols, and foods that sustain microbiome diversity, normobiosis, microbial mediators, and intestinal barrier function. Preserving gut microbial diversity through structured nutrition and stool testing is essential to maintain the anti-inflammatory effects of the microbiome and support long-term metabolic and immune health.
GLP-1 RAs: From Rare to Routine
GLP-1 RA are increasingly prescribed and recognized as therapeutics for metabolic health and cardiovascular health, with benefits that extend beyond weight loss. These benefits must be balanced with their impact on the gut microbiome, as many patients significantly alter their dietary patterns under the influence of these medications. Unstructured caloric restriction may drive gut microbiome shifts that ultimately lead to adverse sequelae.
Best practices include patient education regarding dietary intake, alongside individualized gut microbiome biomarker assessments of diversity, dysbiosis indices, keystone bacterial populations and SCFAs via objective analysis. Strong evidence supports long‑term GLP‑1 RA therapy for a broad patient population, and integrating awareness of gut microbiome shifts can help clinicians sustain therapeutic efficacy while mitigating adverse effects.
Dr. Malkowski will be discussing the GI microbiome at length at the Laboratory, Endocrine, and Neurotransmitter Symposium (LENS) in Las Vegas, February 13-14, 2026.
Attendees can earn up to 14.5 AMA PRA Category 1 Credits, and speakers include Jill Carnahan, MD, Deanna Minich, PhD, Lara Zakaria, PharmD, Mark Tager, MD, and Lylen Ferris, ND.
Save $30 off registration rates with code DRKF30 – and register with a colleague for even bigger savings! Visit fx-ed.com to learn more and register.
Author Bio

Julia Malkowski ND, DC, BSc
Dr. Julia Malkowski enjoys empowering individuals via “docere”; to teach, to delight and to move. Upon experiencing the excellent results of restoring the health of her infant son, Dr. Malkowski began her career in Naturopathic Medicine. Dr. Malkowski graduated with a doctorate in Naturopathic Medicine in December 2017, and a doctorate in Chiropractic Medicine in April 2016 from National University of Health Sciences. Dr. Malkowski has presented at International College of Integrative Medicine (ICIM), the Institute of Women’s Health and Integrative Medicine (IWHIM) and The American Academy of Anti-Aging Medicine (A4M). Dr. Malkowski has been published in Journal of Restorative Medicine, The American College for the Advancement in Medicine and The Townsend Letter. As a licensed physician with over a decade of expertise in functional diagnostics, systems-based education, and clinician mentorship, Dr. Malkowski advances the field of functional medicine as an international speaker, strategic consultant and Staff Doctor in the Clinical Education Department at Doctor’s Data, Inc.
Resources:
- A Gut Microbiome Alternative to Injectables for Weight Loss
- GI360™
- GI360TM Publications
- GI360TM Resource Guide
- GI360TM Sample Report
- A 360-Degree View of the Microbiome: Practical Strategies for Clinicians (Podcast with Dr. Malkowski and Dr. Fitzgerald)
- Exploring Gut Health: Diving into the GI360TM (webinar)
Connect with Doctor’s Data on social:
References:
Mahase E. GLP-1 agonists: US sees 700% increase over four years in number of patients without diabetes starting treatment. BMJ. 2024;386:q1645. doi:10.1136/bmj.q1645
Vahratian, A., & Warren, A. (2025, August). GLP-1 injectable use among adults with diagnosed diabetes: United States, 2024 (NCHS Data Brief No. 537). National Center for Health Statistics, Centers for Disease Control and Prevention. https://dx.doi.org/10.15620/cdc/174616
Alharbi SH. Antiinflammatory role of glucagonlike peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:118. doi:10.1177/20420188231222367
GorgojoMartínez JJ, MezquitaRaya P, CarreteroGómez J, Castro A, CebriánCuenca A, de TorresSánchez A, GarcíadeLucas MD, Núñez J, Obaya JC, Soler MJ. Clinical recommendations to manage gastrointestinal adverse events in patients treated with GLP1 receptor agonists: A multidisciplinary expert consensus. J Clin Med. 2023;12(1):145. doi:10.3390/jcm12010145
Gebrayel P, Nicco C, Al Khodor S, Bilinski J, Caselli E, Comelli EM, Egert M, Giaroni C, Karpinski TM, Loniewski I, Mulak A, Reygner J, Samczuk P, Serino M, Sikora M, Terranegra A, Ufnal M, Villeger R, Pichon C, Konturek P, Edeas M. Microbiota medicine: towards clinical revolution. J Transl Med. 2022 Mar 7;20(1):111. doi: 10.1186/s12967-022-03296-9. PMID: 35255932; PMCID: PMC8900094.
Lauren A Jones, Daniel I Brierley, GLP-1 and the Neurobiology of Eating Control: Recent Advances, Endocrinology, Volume 166, Issue 2, February 2025, bqae167, https://doi.org/10.1210/endocr/bqae167
Gabe MBN, Breitschaft A, Knop FK, Hansen MR, Kirkeby K, Rathor N, Adrian CL. Effect of oral semaglutide on energy intake, appetite, control of eating and gastric emptying in adults living with obesity: A randomized controlled trial. Diabetes Obes Metab. 2024;110. doi:10.1111/dom.15802
Fallows E. Malnutrition with use of GLP1 agonists is an underestimated realworld harm. BMJ. 2025;390:r1512. doi:10.1136/bmj.r1512
Chen, J., Siliceo, S.L., Ni, Y., Nielsen, H.B., Xu, A., & Panagiotou, G. (2023). Identification of robust and generalizable biomarkers for microbiome-based stratification in lifestyle interventions. Microbiome, 11, 178. https://doi.org/10.1186/s40168-023-01604-z
Wastyk, H.C., Fragiadakis, G.K., Perelman, D., Dahan, D., Merrill, B.D., Yu, F.B., Topf, M., Gonzalez, C.G., Van Treuren, W., Han, S., Robinson, J.L., Elias, J.E., Sonnenburg, E.D., Gardner, C.D., & Sonnenburg, J.L. (2021). Gut-microbiota-targeted diets modulate human immune status. Cell, 184(16), 4137–4153.e14. https://doi.org/10.1016/j.cell.2021.06.019
Aliberti, S.M., & Capunzo, M. (2025). The Power of Environment: A Comprehensive Review of the Exposome’s Role in Healthy Aging, Longevity, and Preventive Medicine—Lessons from Blue Zones and Cilento. Nutrients, 17(4), 722.
Gruzdys, V., Malkowski, J., & Mortensen, J. (2025). Gut Microbiota Differences with Age and among North American, European, and Australian Regions. Journal of Restorative Medicine, 15(1), 34–53.
Cong, J., Zhou, P., & Zhang, R. (2022). Intestinal microbiota-derived short chain fatty acids in host health and disease. Nutrients, 14
Peng L, Li ZR, Green RS, Holzman IR, Lin J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMPactivated protein kinase in Caco2 cell monolayers. J Nutr. 2009;139(9):16191625. doi:10.3945/jn.109.104638. PMID: 19625695.
Vinolo MAR, Rodrigues HG, Nachbar RT, Curi R. Regulation of inflammation by short chain fatty acids. Nutrients. 2011;3(10):858876. doi:10.3390/nu3100858. PMID: 22254083.
Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, McGowan BM, Rosenstock J, Tran MT, Wadden TA, Wharton S, Yokote K, Zeuthen N, Kushner RF; STEP 1 Study Group. Onceweekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):9891002. doi:10.1056/NEJMoa2032183
Lincoff AM, BrownFrandsen K, Colhoun HM, Deanfield J, Emerson SS, Esbjerg S, HardtLindberg S, Hovingh GK, Kahn SE, Kushner RF, Lingvay I, Oral TK, Michelsen MM, Plutzky J, Tornøe CW, Ryan DH; SELECT Trial Investigators. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):22212232. doi:10.1056/NEJMoa2307563








